Australian Pulse Bulletin
Faba bean: Integrated disease management
Summary of strategies
- Paddock selection. Separate the current faba bean crop from last year’s faba bean stubble by a minimum distance of 500 m. Reduce disease risk by not sowing adjacent to vetch or lentil stubbles. Aim for a break of at least 4 years between faba bean crops in the same paddock.
- Sowing date. Avoid sowing too early and target the optimum sowing window for your district.
- Variety selection. Select the least susceptible variety to the main disease risk in your region.
- Seed source. Seed retained on-farm should be from the ‘healthiest’ bean crops. Avoid using seed with greater than 10% chocolate spot or 5% ascochyta.
- Inoculation. Inoculate with the correct strain of rhizobia to ensure nodulation, particularly on acidic soils.
- Sowing rate. Aim for a plant population of at least 20 plants/m2 for faba bean, at least 15 plants/m2 for small broad bean and larger sized faba bean, and at least 8 plants/m2 for larger broad bean.
- Foliar fungicide application. Success of the fungicide program is dependant on monitoring, correct disease identification and timeliness of spraying with the right fungicide.
- Harvest management. Harvest early to minimise disease infection on seed. Consider windrowing or desiccation as a tool to enable earlier harvesting.
Diseases of faba bean
The major fungal diseases affecting faba bean grown in the southern region are chocolate spot (Botrytis fabae), ascochyta blight (Ascochyta fabae) and cercospora leaf spot (Cercospora zonata). Rust (Uromyces viciae-fabae) can also become a problem in prolonged wet seasons. These diseases can be effectively controlled to prevent them from causing yield loss. However, management decisions and seasonal conditions play a significant role in disease outbreaks and grain yield. Bean growers need to implement an integrated approach to disease management in most years to produce a profitable crop.
Symptoms of ascochyta and chocolate spot, the most damaging diseases, can be confused with symptoms of cercospora leaf spot. In addition, symptoms may be similar to damage on leaves from herbicides or physical events, which then allow minor diseases such as alternaria (Alternaria alternata) to infect the plant tissue.
Correct disease identification is necessary to avoid unnecessary spraying or incorrect fungicide use.
Chocolate spot is favoured by warm (15–25°C), humid conditions (over 70% RH) that extend for 4–5 days. Under such conditions the disease can spread rapidly within a crop. It typically develops later in the season during flowering and after canopy closure. Yield loss due to chocolate spot results from pod abortion and plant damage.
Ascochyta infection is likely to occur in environments with prolonged wet, cool (5–15°C) conditions and usually develops early in the growing season. Damage from stem infection often results in serious crop lodging in susceptible varieties. However, development of this disease can also be important late in the season. Pod infection with ascochyta blight can cause seed staining and the subsequent downgrading of faba bean grain.
Cercospora, like ascochyta, develops early in the season during wet and cold conditions, but is less damaging to the crop. The disease can develop later in the season, similarly to chocolate spot, and if uncontrolled, can cause extensive defoliation.
Rust can occur from mid spring onwards and is favoured by warm temperatures (above 20°C). Rust infection can occur following 6 hours of leaf wetness, so does not require extended wet periods.
-

Ascochyta blight pod lesions affect seed quality (DPI Vic). Treatment is too late when it gets to this stage.
-

Ascochyta blight leaf lesion (SARDI).
-

Bean grain with ascochyta blight damage (SARDI).
-

Cercospora (SARDI).
-

Chocolate spot on leaves and flowers (SARDI).
-

Chocolate spot (SARDI).
-
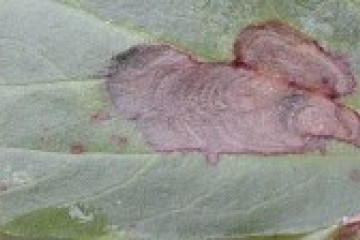
Chocolate spot leaf lesions (SARDI).
-

Chocolate spot lesion on pod leading to infection and staining on seed (SARDI).
-

Poor pod set and leaf loss from failing to protect against chocolate spot early (Pulse Australia).
-

Bean rust (SARDI).
-

Spotting from herbicide application (SARDI).
-

Bean leaf roll virus (BLRV). Note bare ground around the plants (Pulse Australia).
-

Bean seed showing Pea Seed-borne Moasaic Virus (PSbMV) marking (SARDI).
-

Weather stained faba beans (SARDI).
On-farm hygiene
(post harvest-pre sowing)
Chocolate spot and ascochyta can carry-over from one season to the next on infected faba bean seed, stubble and volunteer plants. Seed retained on-farm should be from the healthiest crop, where disease was not detected.
Undertake a program of stubble reduction i.e. burn or bury infected crop residue.
Grazing over summer helps reduce stubble, however, be aware that stock can carry infected stubble between paddocks. Be aware of grazing restrictions on stubble from crops treated with fungicides.
Infected stubble may also be carried by wind, water (particularly flooding) or machinery at harvest. Where practical, clean all machinery, transport equipment and storage bins thoroughly with compressed air before moving to the next paddock.
Control volunteer faba bean, vetch and lentil seedlings early to limit the build-up of disease inoculum.
Paddock selection
Avoid sowing adjacent to faba bean stubble, particularly downwind. If possible, aim to separate the current faba bean crop from last year’s bean stubble by a minimum distance of 500 m.
A break of at least 4 years between faba bean crops is recommended.
Growers who plan to sow more than one variety of faba beans should ensure there is at least 500 m between different varieties. Faba beans cross-pollinate, increasing the risk of disease resistance breaking down and producing mixed seed types that are difficult to market.
Reduce the disease risk by not sowing adjacent to lentil and vetch crops or stubble as these may harbour Botrytis fabae the primary cause of chocolate spot in faba bean. If this is not possible, employ a management strategy appropriate for situations where there is a high risk of disease.
Ensure the maximum plant-back period for herbicides are adhered to, particularly for sulfonylurea and clopyralid, as herbicide residue may cause significant crop damage and weaken the plant’s resistance to disease.
Varieties
All newer bean varieties released for southern Australia are resistant to ascochyta (PBA Rana, PBA Kareema, Nura and Farah). Older varieties (Fiesta VF, Manafest and Fiord) and varieties released for northern Australia (Doza, Cairo) are susceptible to ascochyta blight. Under extreme disease pressure, ascochyta blight can still develop in a resistant variety.
Sow an ascochyta resistant variety in districts where ascochyta has been a continual problem. There is unlikely to be a yield penalty with ascochyta resistant varieties compared with a susceptible variety when ascochyta blight is either absent or controlled.
Doza and Cairo should not be grown in southern NSW, Victoria or South Australia. They are susceptible to ascochyta and chocolate spot.
Nura and Farah are moderately susceptible and susceptible to chocolate spot, so fungicide protection is still necessary in wetter areas or springs.
Nura and PBA Kareema are the only southern varieties with a degree of resistance to rust.

Because no variety has resistance to all of the faba bean diseases, it is important to select the correct fungicide and application timing to best manage the target disease in the chosen variety. Control strategies vary according to the variety being grown.
Seed
Avoid using seed with greater than 10% chocolate spot or 5% ascochyta infection, and where possible use seed with nil infection. Seed-borne inoculum is usually less important than stubble-borne inoculum. However, infected seed can be important in the establishment of either disease in new faba bean growing regions.
Weathering of crops after ripening can substantially reduce the level of germination in harvested seed. Poor storage of seed can also significantly reduce the level of germination.
If seed is more than one year old, frosted or weather damaged, its germination and vigour may have deteriorated in storage.
SARDI Diagnostic Services
Phone: 08 8303 9360
http://pir.sa.gov.au/research/services/crop_diagno...
Seed dressing
The presence of rhizobium for effective nodulation and healthy plant growth is usually more important than a fungicidal seed treatment. Use of granular inoculant or fluid injection of rhizobia avoids contact between rhizobia and any seed treatment.
Rhizobium strain WSM 1455 (Group F) is the faba bean inoculant suited to all soil types.
Thiabendazole plus thiram (eg P-Pickel T®) is registered in faba bean for seedling root rots (Fusarium spp. and Pythium spp.).
Sowing rate
Higher plant populations can exacerbate foliar disease development. Avoid sowing headlands. Sowing at lower plant populations sometimes reduces the risk of foliar disease, as the crop is usually less bulky. However, seeding rates resulting in lower than recommended plant populations could reduce potential crop yield.
Optimum plant populations for most faba beans (e.g. 50–70 g/100 seeds) can range from 20–35 plants/m2.
For the medium to larger sized faba beans and for small broad bean seed (e.g. 85-110 g/100 seeds), optimum plant populations can vary from 15–25 plants/m2.
For larger broad bean seed (e.g. 110-160 g/100 seeds), optimum plant populations can vary from 8–12 plants/m2.
Row spacing and sowing
Faba bean row spacing varies from 15–25 cm, but up to 55 cm with broad bean.
For faba bean, using wider row spacing (25-60 cm) may assist with control of chocolate spot by delaying canopy closure and reducing humidity between rows.
Some growers are now using row spacing of 75–100 cm, or using paired rows, to improve pod set, delay canopy closure and hopefully minimise disease. Wide rows could also lead to crop lodging, which can exacerbate disease. Wide rows do, however, allow herbicides to be sprayed between rows with hooded shields or targeted row spraying with fungicides to reduce the amount of product applied.
To avoid potential ‘hotspots’ for disease, consider ‘tram lining’ and controlled traffic. Physical damage to the crop from machinery travelling over the paddock can be a major cause of disease outbreaks.
Some growers believe that sowing in wide rows in a north-south direction improves pod set and disease control.
Sowing date
Early sowing may result in a bulky crop, which creates an environment most conducive to foliar disease.
Later sowings reduce disease risk but can result in lower yields due to the risk of dry conditions and high temperatures at flowering to pod fill. Yield decreases of 150-250 kg/ha are likely for every week past the optimum sowing time.

Soil type and nutrition
Manage the beans to minimise stress from soil type or nutritional problems. Otherwise disease needs to be well managed because of the greater risk, despite less vegetative growth being produced. Manage soil chemical constraints such as:
- If soils are of low pH, inoculate the beans and ensure good nodulation.
- Mn/Al toxicity can impact on soils of low pH.
- If soils have inadequate P and/or Zn levels, apply suitable fertiliser.
- Manage the beans for Fe/Mn deficiency on soils of high pH.
- Avoid or manage soils that waterlog or have a sub-soil compaction layer.
- Potassium deficiency can occur under wet, cold conditions on some low pH or lighter textured soils, leading to shorter plants, loss of lower leaf and greater susceptibility to foliar disease.
Virus incidence and control
In some seasons, viruses can become a problem in crops. Viruses such as pea seed-borne mosaic (PSbMV), bean leaf roll virus (BLRV) and beet western yellows virus (BWYV) are not seed transmitted but become established after aphid-vector activity.
The most important factors that predispose pulse crops to severe virus infection are:
- Infected seed or close proximity to a substantial virus reservoir (e.g. lucerne, summer weeds, field peas for PSbMV).
- High summer-autumn rainfall and the subsequent uncontrolled multiplication of aphids on host plants. Early aphid flights to newly emerged crops can cause early infection and economic loss as infected plants act as a reservoir for further spread of infection within the crop.
Virus risks can be managed by combining a number of different control measures including:
- Suppress the virus source within the crop. Sow seed with less than 0.1% seed infection.
- Control volunteer weeds during summer and autumn.
- Use a seed treatment of Gaucho 350SD (imidacloprid), which is registered for early aphid protection.
- Ensure faba bean plants are less attractive to aphids by minimising seedling disease, herbicide damage or poor nutrition.
- Decrease aphid landing rates by having stubble cover and/or a dense early canopy.
- Note that high seeding rates and narrow row spacing to provide early canopy closure assists in aphid control, but conflicts with management of fungal diseases.
Growers should only consider applying insecticide for virus control if they consider their crops to be at high risk. Insecticides aimed at controlling damage from aphid feeding are normally too late to control virus spread and damage.
Fungicide program
Check that fungicide registration or permits are current. There are three critical periods for monitoring bean crops for disease:
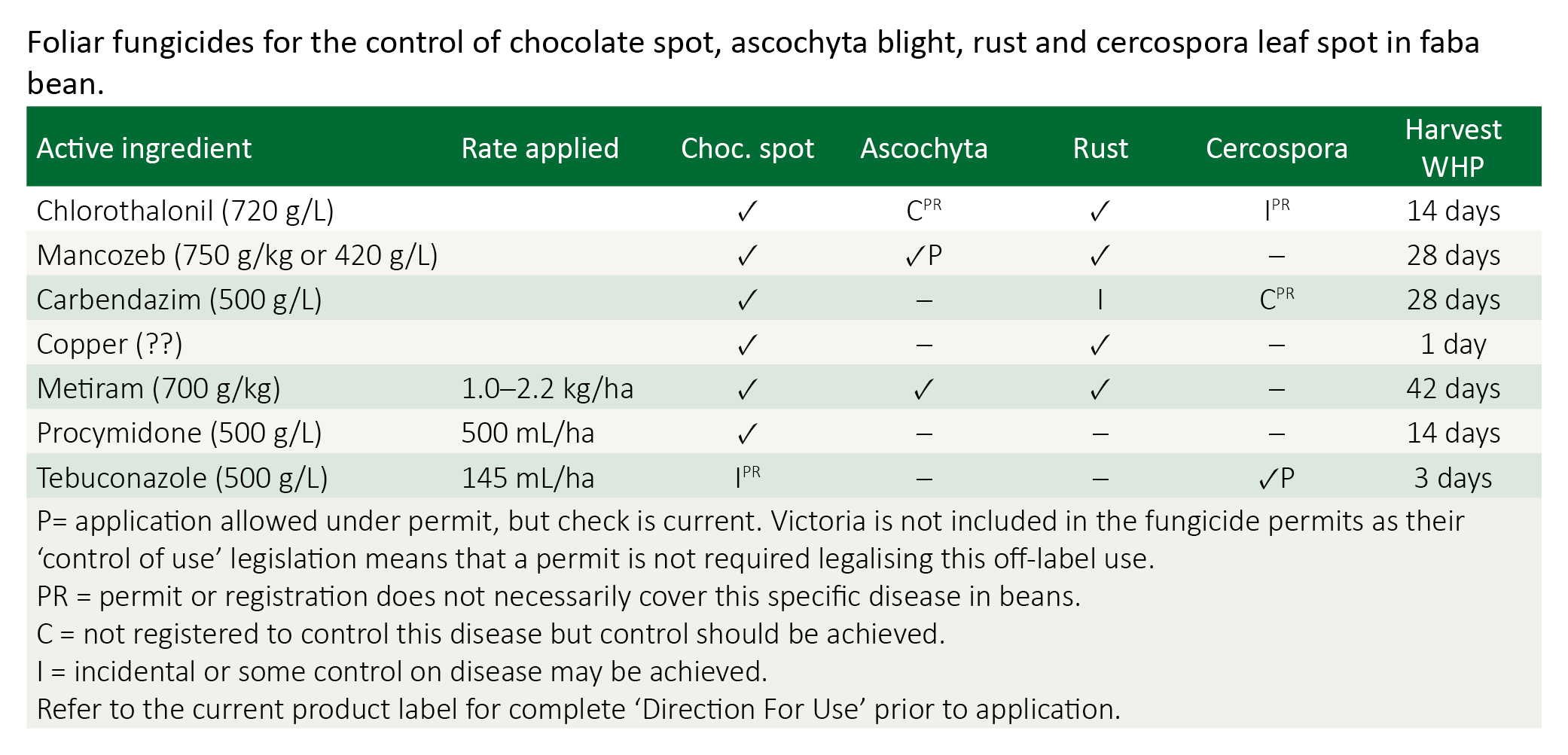
First critical period is 5-8 weeks after emergence. Cercospora leaf spot is most effectively controlled at this period. However, ascochyta blight should be targeted for susceptible varieties like Fiesta VF and Fiord. Fungicide choice depends on the target disease. For control of cercospora, tebuconazole or carbendazim is recommended. For varieties that are susceptible to ascochyta, mancozeb or chlorothalonil are recommended and applied at this period irrespective of symptoms being present.
Second critical period is during flowering (13–16 weeks after emergence) for protection against disease prior to canopy closure. This is a critical period to allow good penetration of fungicide deep into the canopy and to protect lower leaves at a time when disease can impact on yield by causing leaf lesions, flower drop and pod abortion.
Chocolate spot is most devastating at flowering and pod set, so a protective foliar fungicide application at early to mid-flowering, before symptoms appear, is recommended. Ascochyta should again be targeted in susceptible varieties if necessary. A fungicide application to target cercospora may be considered if early control was inadequate. Apply the appropriate fungicide(s) if the crop has sufficient yield potential when:
- rain events are likely
- canopy closure is about to occur
- chocolate spot is present
- ascochyta is present and the variety is susceptible
Mixtures of fungicides may be required to achieve control of multiple diseases in some varieties or situations. Additional spray applications may be required in high disease pressure situations.
Crops are at high risk to chocolate spot if one or more of the recommended practices to reduce disease are not followed, and when:
- sown early, producing bulky canopy
- sown in a high rainfall area
- spring rains are forecast
- disease has established in the lower canopy
Third critical period is at the end of flowering when pods are filling (15-20 weeks after emergence).
Chocolate spot must be controlled when conditions are favourable to the disease. It may also be necessary to control rust if it has become established, as epidemics can form rapidly. The control of ascochyta is also important in susceptible varieties. To control the target disease, apply an appropriate fungicide to protect the crop if it has sufficient yield potential and when:
- rain is forecast
- significant growth has occurred since previous fungicide application(s)
A mixture of fungicides may be required to give control of multiple diseases in some varieties or situations. Seed staining from ascochyta is a major problem when wet weather persists up to harvest. However, some seed staining caused by weathering and delayed harvest will not be reduced by fungicide sprays. Fungicide can be applied until just prior to the crop drying down, but be aware of withholding periods.
During all critical periods, monitor crops at least once every week and observe weather forecasts with impending rain. Visible lesions only appear several days after wet conditions.
Fungicide application during critical periods is a standard practice in high rainfall regions, irrigation districts, in a wet year or in high disease risk situations. A crop is at high risk if susceptible varieties are grown, crop rotation is short, sown adjacent to faba bean stubble, infected seed is sown or where all preventative management strategies cannot be followed. Additional fungicides may be required, particularly if conditions are conducive to disease.
Read the fungicide label or permit before use, and check that the permit is current. Not all products are registered or have permits in faba bean in all states, or for the minor disease they may control while targeting the major disease. Incidental control may be achieved.

There are a range of foliar fungicides for potential use in faba and broad beans.
Controlling cercospora
Cercospora must be controlled early (5–8 weeks). To prevent cercospora disease establishing in a crop:
- Apply tebuconazole early (APVMA permit), or
- Use carbendazim. When applied for the control of other diseases, it can also control cercospora and help retain lower leaves in the canopy.
- Note that chlorothalonil is less effective than carbendazim, but when used for the major diseases can also help to control cercospora.
- Mancozeb does not control cercospora.
Carbendazim may be required later for chocolate spot control, so the maximum allowable applications of this fungicide should be considered.
Controlling ascochyta
If ascochyta risk is high, or it persists and continues to spread in the bean crop, then chlorothalonil is considered more effective than mancozeb. Beware of the grazing with holding period and the export slaughter interval (ESI) restriction with chlorothalonil (63 days). Take note of the withholding period for grain prior to harvest for mancozeb (30 days) and chlorothalonil (14 days).
Ascochyta is targeted in all critical periods, particularly in susceptible varieties.Controlling chocolate spot
Choice of products available includes those containing mancozeb, chlorothalonil, carbendazim, copper or procymidone. Chocolate spot is targeted in critical period 2, as well as critical period 3.
If chocolate spot pressure is high or the disease is spreading in the crop, then carbendazim or procymidone are more effective than chlorothalonil, mancozeb or copper.
Label regulations limit carbendazim to a maximum of two consecutive sprays at 14-day intervals. Carbendazim is a systemic fungicide with single-site specificity so the probability of resistance developing increases with regular use. Alternate carbendazim with either chlorothalonil or mancozeb. Observe the withholding period for grain prior to harvest for carbendazim (30 days).
Controlling rust
Rust can become an important disease in beans with some varieties or seasons during critical period 3. A foliar spray of mancozeb, chlorothalonil or a copper product will control rust.
Mancozeb or chlorothalonil provide the added key benefit of suppressing chocolate spot and ascochyta.
Controlling alternaria
There is no fungicide efficacy data for Alternaria on faba beans, and no product is registered to control it. This disease may be controlled when foliar fungicides are applied to control other diseases.
Fungicide spraying
Control is based on preventing new infections rather than curing old infections.
Fungicide protection can be for 2–3 weeks but may be reduced to 7–14 days during warm periods of rapid growth and intense rainfall or conditions conducive to disease development. Any new growth after fungicide is applied is not protected.
Waiting till after a rain event allows some fungal infection to occur, which then becomes a source of further disease spread.
Spraying in light rain is preferable to waiting till after the end of a rain event. Spraying before the canopy closes achieves better fungicide penetration and is more effective than trying to halt an epidemic after the canopy closes or the crop lodges.
Use high water rates (preferably 100 L/ha by ground or 30 L/ha by air).
For fungicide application choose correct nozzles, droplet spectrum, volumes of water and operating pressure (e.g. 400 kPa). Match nozzle size to speed of travel and desired water volume.
Surfactants (spreaders) improve initial spray deposits and redistribution (refer to fungicide label). Stickers reduce droplet spread over the leaf surface.
Spray early in the morning when dew is present to assist in spread of fungicide over the plant surface.

Disease risk is heavily influenced by the variety grown, the location and season. Fungicide options chosen need to account for the severity of the risk, disease presence or protection requirement.
Early harvest
Harvest at maturity to minimise ascochyta and chocolate spot infection on seed. Seed infection is usually more severe when crops are harvested late. Harvest losses and downgrading in quality can be substantial if bean harvest is delayed until moisture content is below 12%.
Ascochyta can grow on dead plant tissue once wet, so infection on pods can grow through onto the seed if harvest is delayed and conditions have been wet.
Consider windrowing or desiccation as a tool for early harvesting.
Windrowing does not necessarily eliminate weathering damage of beans, particularly if harvest is delayed.
Bean quality is critical for marketing. The main grade for faba bean receival standard, referred to as Australian No.1 Grade, allows 8% maximum defective beans, inclusive of 3% maximum poor colour (due to disease, water staining, frost etc). A second, lower grade receival standard, Australian No.2, may be segregated at some silos, allowing up to 10% defective beans, inclusive of 7% maximum poor colour.
Acknowledgements
Wayne Hawthorne, Pulse Australia, Rohan Kimber and Jenny Davidson, SARDI, Jeff Paull, University of Adelaide, Jason Brand and Helen Richardson, DPI Victoria. Also, Kris Panagiotopoulos, formerly of Pulse Australia, Trevor Bretag, formerly DPI Vic., Mary Raynes, DPI Vic., Alex Nikandrow, formerly NSW Agriculture and Peter Matthews, formerly NSW DI&I
Key contacts
Disclaimer
Information provided in this guide was correct at the time of the date shown below. No responsibility is accepted by Pulse Australia for any commercial outcomes from the use of information contained in this guide.
The information herein has been obtained from sources considered reliable but its accuracy and completeness cannot be guaranteed. No liability or responsibility is accepted for any errors or for any negligence, omissions in the contents, default or lack of care for any loss or damage whatsoever that may arise from actions based on any material contained in this publication.
Readers who act on this information do so at their own risk.
Copyright © 2015 Pulse Australia
All rights reserved. The information provided in the publication may not be reproduced in part or in full, in any form whatsoever, without the prior written consent of Pulse Australia.
Last updated: 15 January 2016
